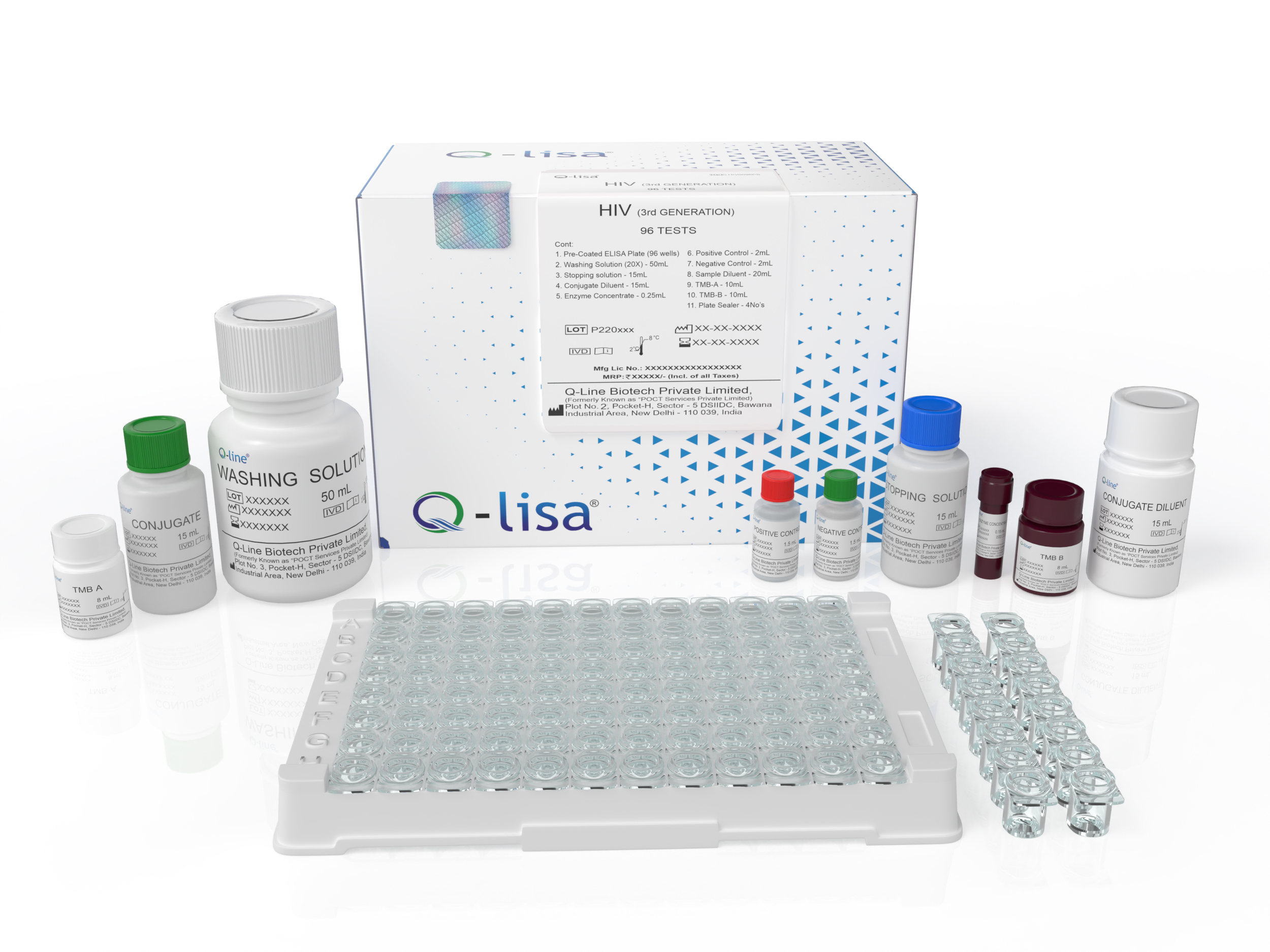
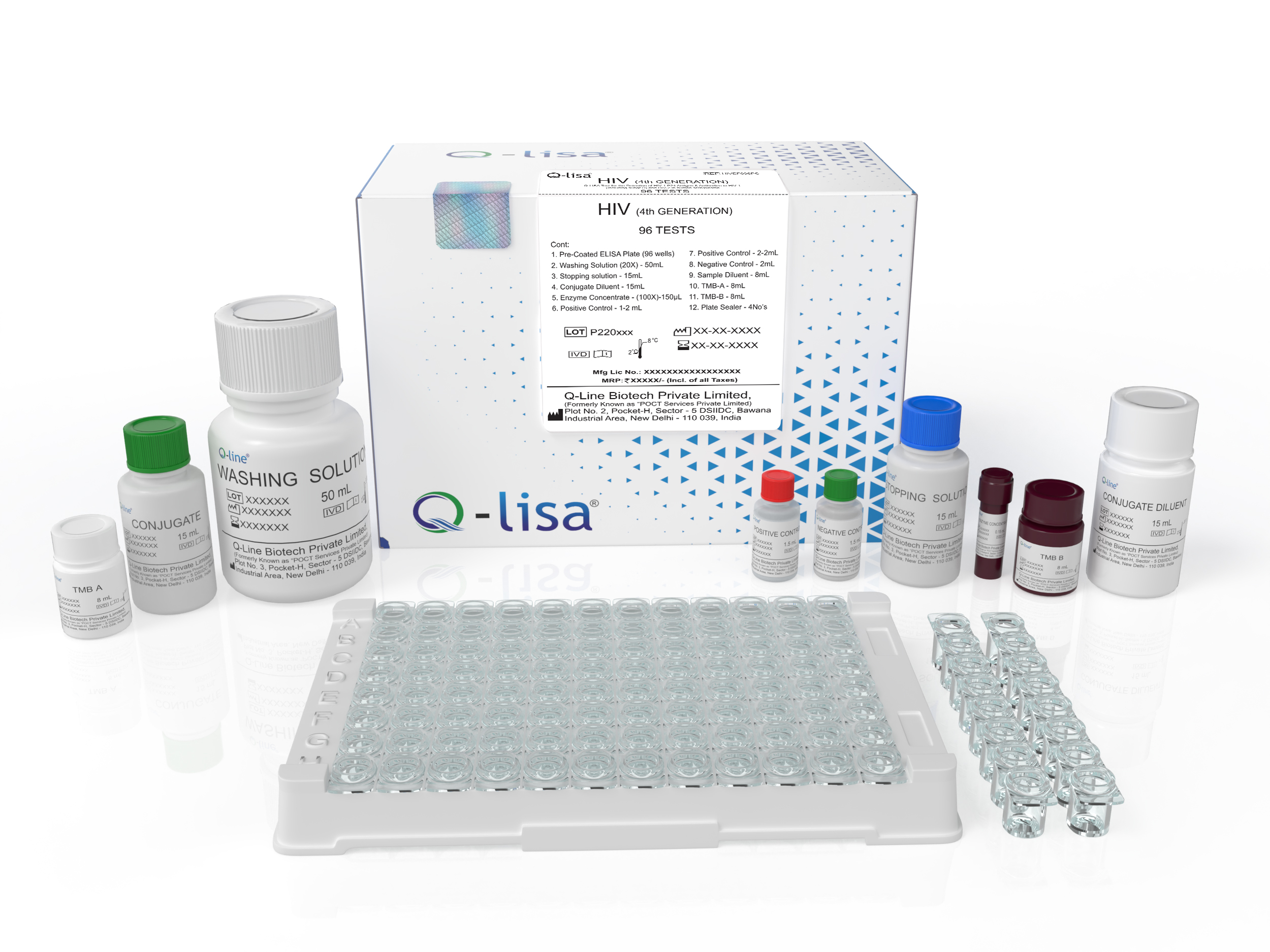
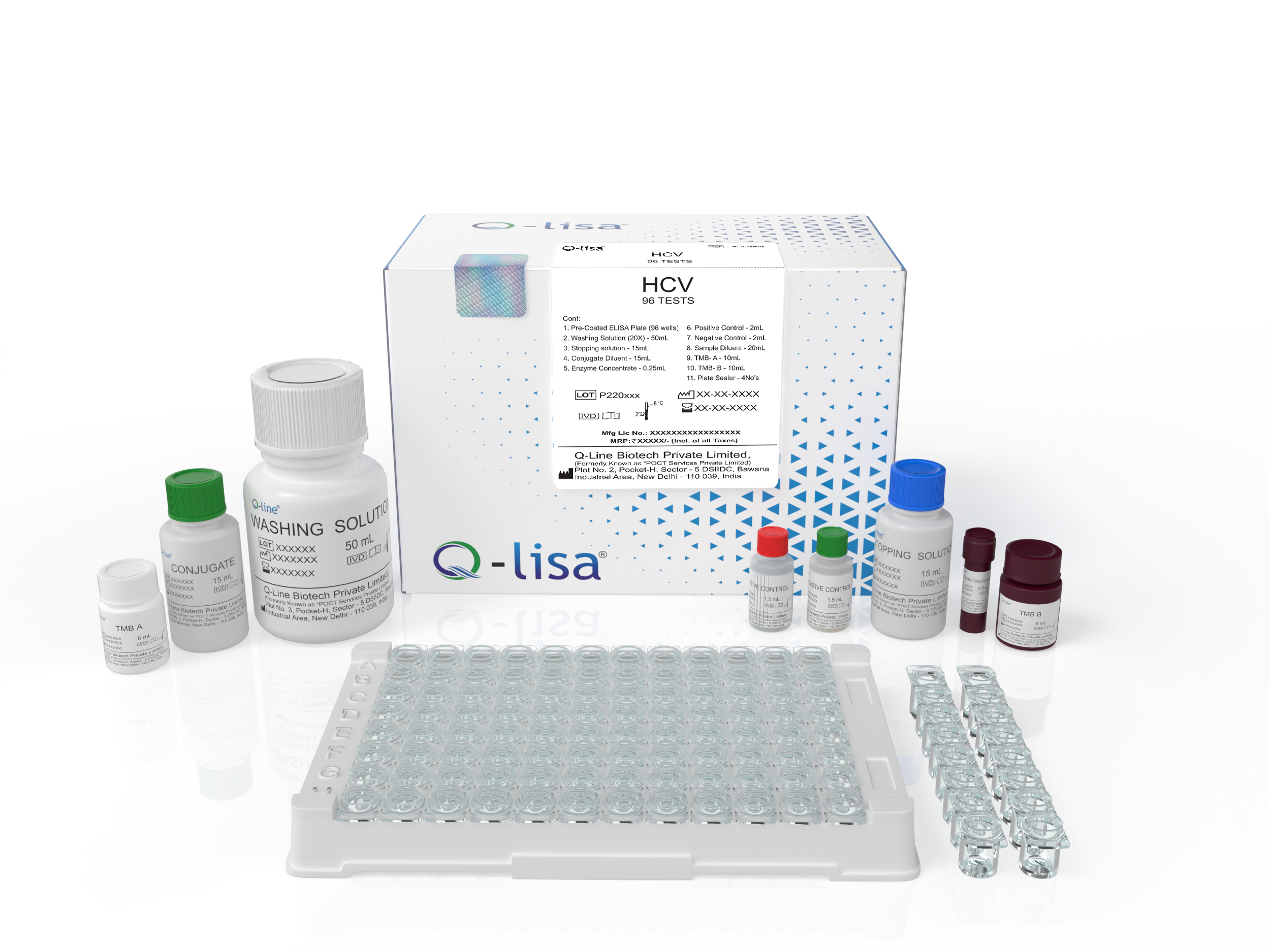
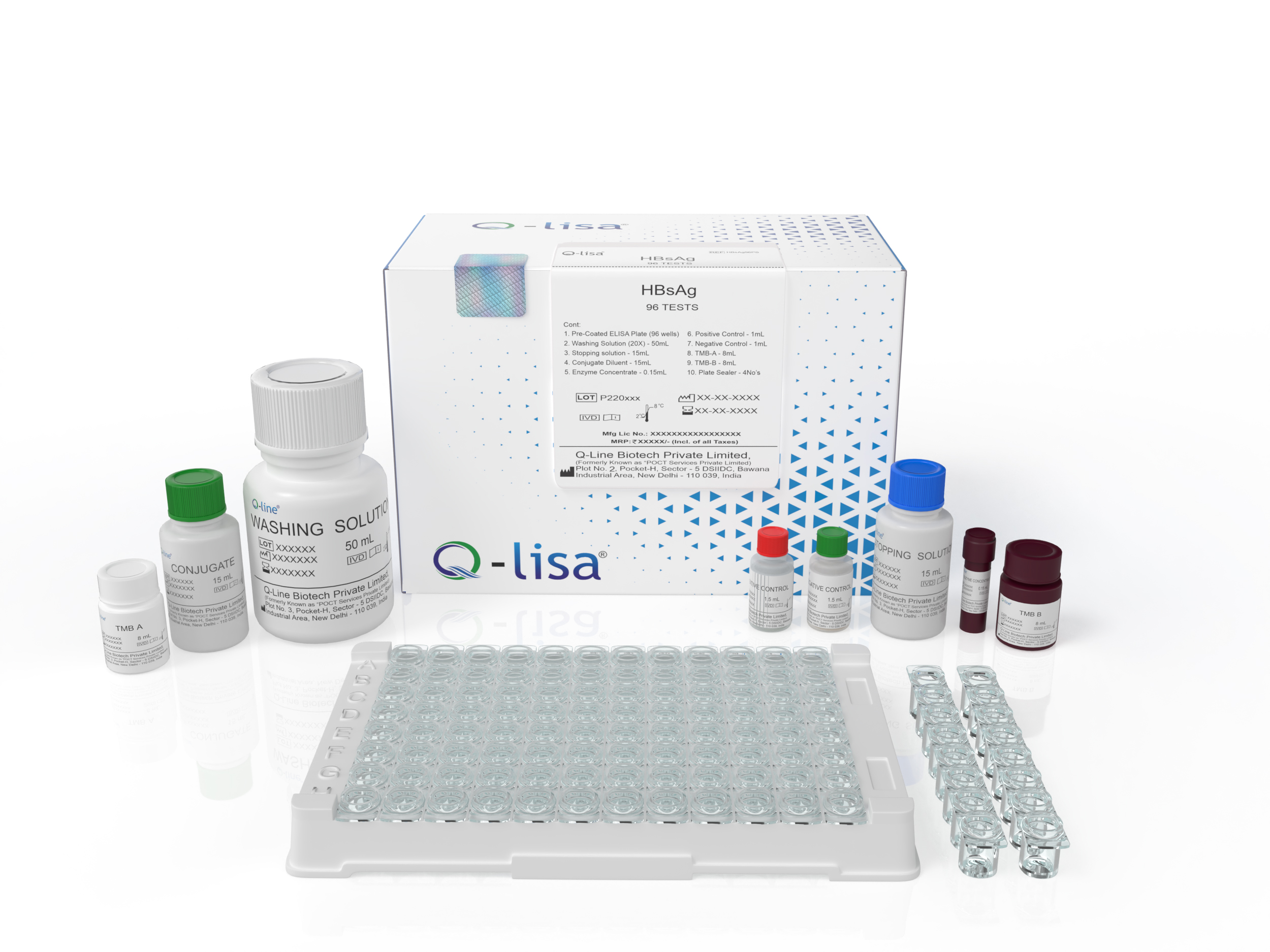
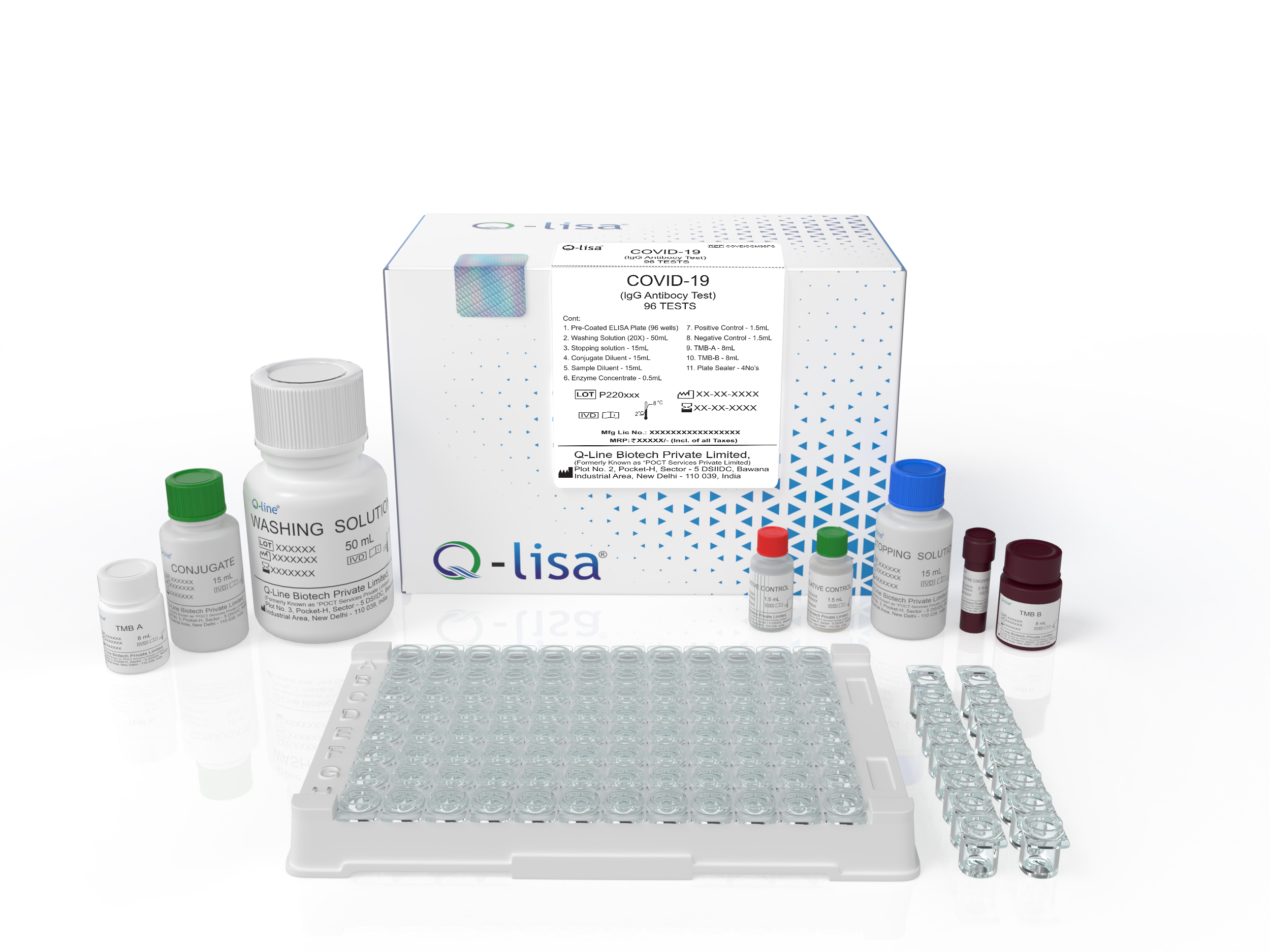
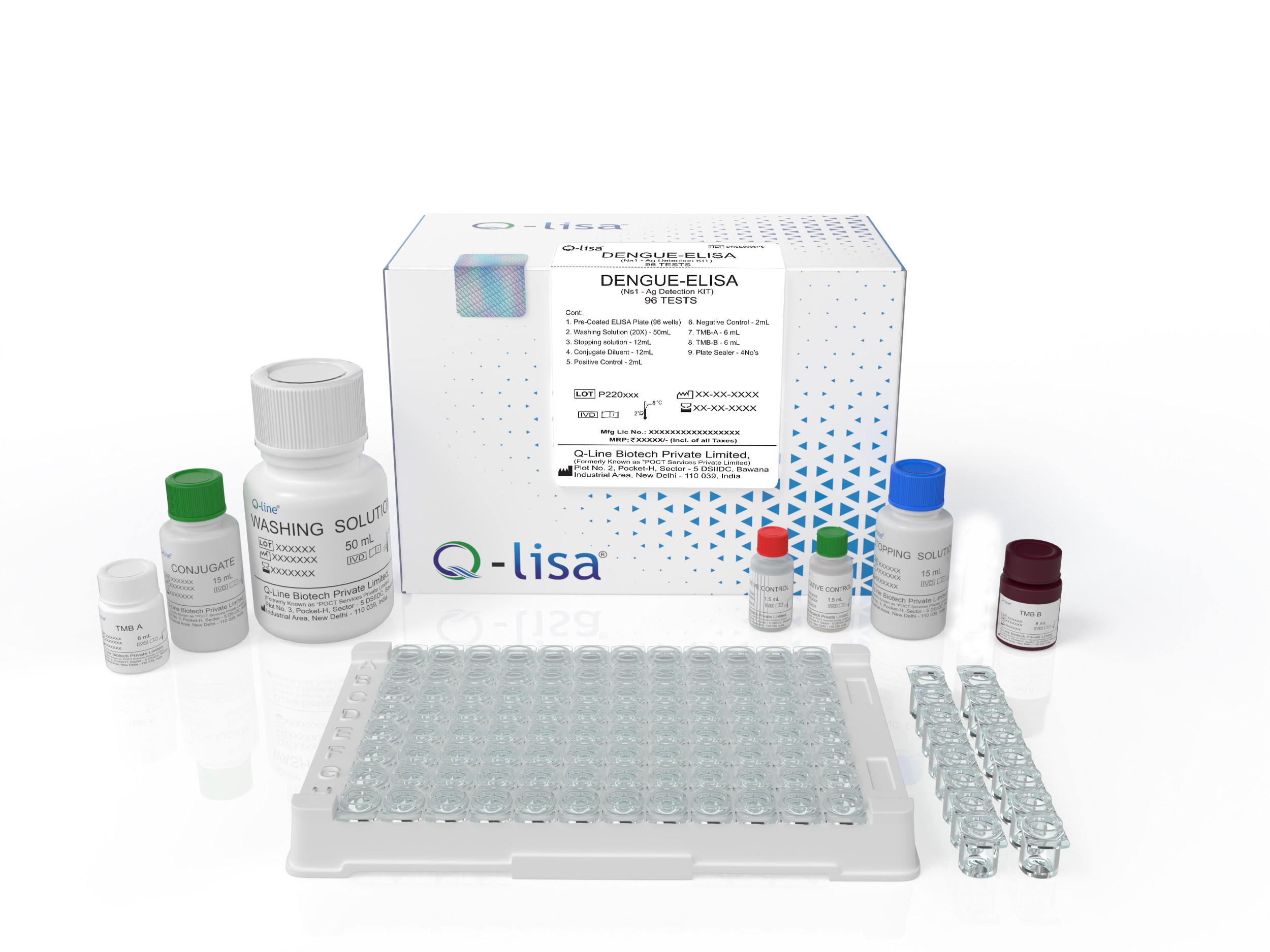
Product Description
Available Options:
| REF | Volume |
|---|

| REF | Volume |
|---|


We use cookies to improve user experience, and analyze website traffic. For these reasons, we may share your site usage data with our analytics partners. By continuing to the site, you consent to store on your device all the technologies described in our cookie policy. Here is the the cookie policy
